1.1 What is Diffusion?
The molecules in a gas are in constant, chaotic thermal motion. Because the molecules are free to move about, they continually intermingle and collide with one another. While the kinetic energy of individual molecules is variable, the average kinetic energy of molecules in a gas at uniform temperature is constant, even if the gas is made up of multiple species with different molecular masses. Massive molecules move more slowly than do lighter molecules by just the amount necessary to make the kinetic energies equal on average.
Because the average kinetic energies of all constituent species are the same in a gas at constant temperature, a non-uniform distribution of constituent species as indicated by variable concentration does not result in a variable distribution of kinetic energies for the gas as a whole. On the other hand, variable constituent concentration (i.e., variable number of molecules per unit volume) means that the average kinetic energy of individual components is correspondingly non-uniform. Molecules of individual constituents migrate from positions where the local average constituent kinetic energy is high to positions where it is low. This migration is diffusion.
The intermingling of two gases in a diffusion chamber located between supply headers at the same pressure is depicted in Figure 1a. Because there is no pressure difference across the chamber between headers A and B, only diffusion is responsible for any transport that occurs. As gases A and B intermingle (diffuse into one another) they form a single gas with two components. The mixture is referred to as a binary gas. The constituents of the binary gas diffuse from their respective sources in the direction of decreasing concentration. The diffusive motion is the translation of an aggregate of molecules and may be expressed as a rate of flow measured in moles, volume, or mass per unit time.
Suppose we were to conduct an experiment in which steady diffusion of a binary gas at uniform temperature and pressure occurs in this chamber that connects headers at identical pressures (Figure 1a). First, consider the case in which the diffusion chamber is devoid of solid particles. In this case, the only impediment to diffusion of each constituent is that which arises from the collision with molecules of the other constituent (collisions with the walls of the large diameter chamber are neglected). It is diffusion of this kind that is most commonly treated in text books (e.g., Bird et al., 2002; Cussler, 1997) and for which Fick’s law (Fick, 1855) applies (see Section 2.4 for a presentation of Fick’s law). Both experimental observation and Fick’s law show that the two components of the binary gas diffuse into one another at equal rates and in opposite directions, even though the molecular weights may be different. Thus, the diffusive fluxes of the constituents mutually cancel and the binary gas, as a whole, experiences no net motion. This kind of diffusion is often referred to as ordinary diffusion.
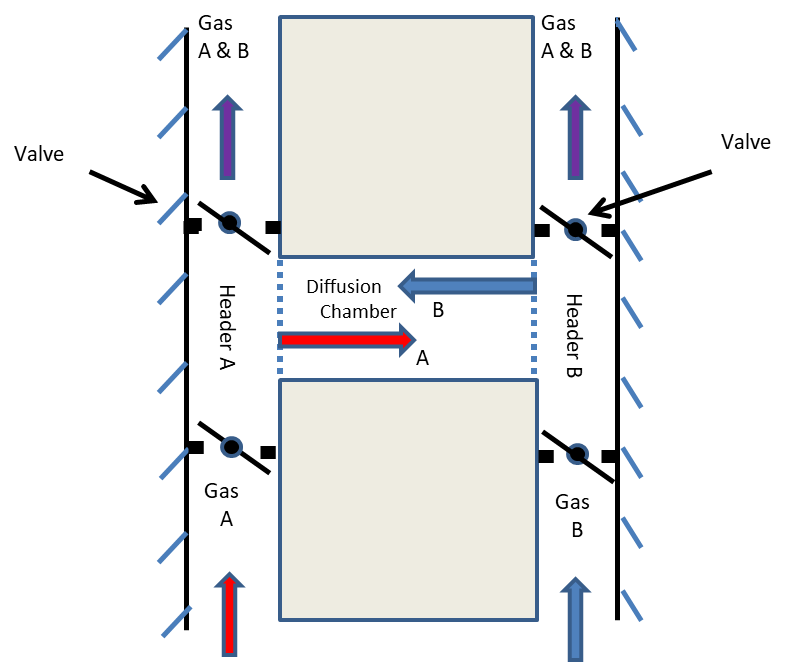
Figure 1a – A thought experiment for steady binary diffusion between headers at identical pressures. Gas A is introduced in the left-hand header, diffuses through the chamber toward the right, and is exhausted in the mixture at the top of the right-hand header. Gas B, introduced in the right-hand header, diffuses toward the left, and is exhausted as part of the mixture at the top of the left-hand header. The concentrations of gas A in the exhaust from the right header and of gas B from the left header are minimized by large flow rates in the headers. Gases A and B diffuse at the same rates and in opposite directions.
Now consider a second experiment in which diffusion occurs in the chamber after it was filled with sand (Figure 1b). An obvious effect of filling the diffusion chamber with sand is that the cross-sectional area available for gas-phase diffusion is reduced. Also, the sand causes the diffusion path to be tortuous and, therefore, longer than in the first experiment. These obstructions equally effect diffusion of both components and do not account for the surprising fact that the diffusion rates of the two constituents are generally observed to be of unequal magnitude and no longer mutually cancel. One manifestation of the unequal diffusion rates is a bulk flow of the binary gas in the direction of diffusion of the component with the lower molecular weight. This diffusion-generated bulk flow occurs even though there is no pressure gradient. The commonly held notion that viscous flow and flow of the fluid as a whole are synonymous is not true for binary gases in a porous medium.

Figure 1b – Steady binary diffusion in a particle-filled chamber. Gas A has a lower molecular weight and diffuses more rapidly to the right than does gas B to the left, as indicated by the arrows.
Yet another unexpected feature of gas diffusion in porous media is the coupling that exists between diffusion and viscous flow. Should all the valves in the apparatus shown in Figure 1b be simultaneously closed at some point during the second experiment, a pressure gradient develops that drives bulk gas flow in the direction opposing the diffusion-generated bulk flow. This feature of diffusion does not occur if the diffusion chamber is free of solid particles and wall effects are negligible. This is another manifestation of how the presence of solid particles affects diffusion in profound and fundamental ways.
