3.1 Investigating Groundwater Mixing
Conservative tracers can provide quantitative information on mixing between different water sources. When two or more water sources (often termed end-members) that have different isotopic or chemical compositions are mixed, then the chemical composition of the mixture will depend upon the composition of each end-member and the proportion of each end-member in the mixture. Thus, Equation 7 can be written for mixing of two water sources.
| cm = c1f + c2 (1 – f) | (7) |
where:
| cm | = | concentration of the mixed water |
| c1 | = | concentration of conservative tracer in end member water 1 |
| c2 | = | concentration of conservative tracer in end member water 2 |
| f | = | proportion of end member 1 in the mixture |
| 1 – f | = | proportion of end member 2 in the mixture |
Here, the concentrations must be absolute concentrations, and not isotope ratios, as discussed below. Thus, if the concentrations in the two sources (end-members) are known (or assumed), and their concentrations in the mixed sample are measured, then Equation 7 can be re-arranged into Equation 8 and hence the proportions of each end-member in the mixture can be determined.
| (8) |
Although concentrations of a single conservative tracer only need be measured to calculate proportions of two end-members in a mixture, it is more common to measure concentrations of more than one tracer. This will give increased confidence in results of the mixing calculations. When two different tracers are measured, then a mixing plot can be constructed showing the concentrations of the two end-members in the mixture, and the concentration of the mixed sample.
The concentration of a conservative element in a mixture of two end-members will lie on a straight line between these end-members (Figure 11). This straight line is often called a mixing line. Where several samples fall on mixing line, then this is evidence that they may be the products of mixing. However, if the ratios of elements, or isotope ratios (e.g., 87Sr/86Sr, 13C/12C), are plotted then the mixing line can be curved rather than straight. In this case, Equation 7 will also not be correct. This occurs most frequently when an isotope ratio is plotted against the total concentration of the element (e.g., 87Sr/86Sr ratio versus total Sr concentration). It occurs because at higher element (e.g., Sr) concentrations, a larger change in isotope concentration (e.g., 87Sr concentration) is required to change the isotope ratio than at lower element concentrations (Section 2.5.3 of Kendall and Caldwell, 1998).
A similar approach can be applied where there are more than two end-members, although more than one conservative tracer will be needed. The mixing equation for ‘n’ end-members is shown as Equation 9.
| (9) |
where:
| cAm | = | concentration of tracer A in mixture |
| cAi | = | concentration of tracer A in end member water n (same units as cAm) |
| fn | = | proportion of end member ‘n’ (dimensionless) |
In this case there are ‘n’ proportions: f1, f2, …, fn. However, since the proportion of the last end-member, fn, can be calculated from the other proportions (i.e., fn = 1 – f1 – f2 – … – fn–1), there are only n-1 unknowns. To solve this equation, n-1 equations are needed, which means that n-1 different conservative tracers need to be measured. The equations for the different tracers can then be solved simultaneously to determine the proportions of the ‘n’ different end-members in the mixture.

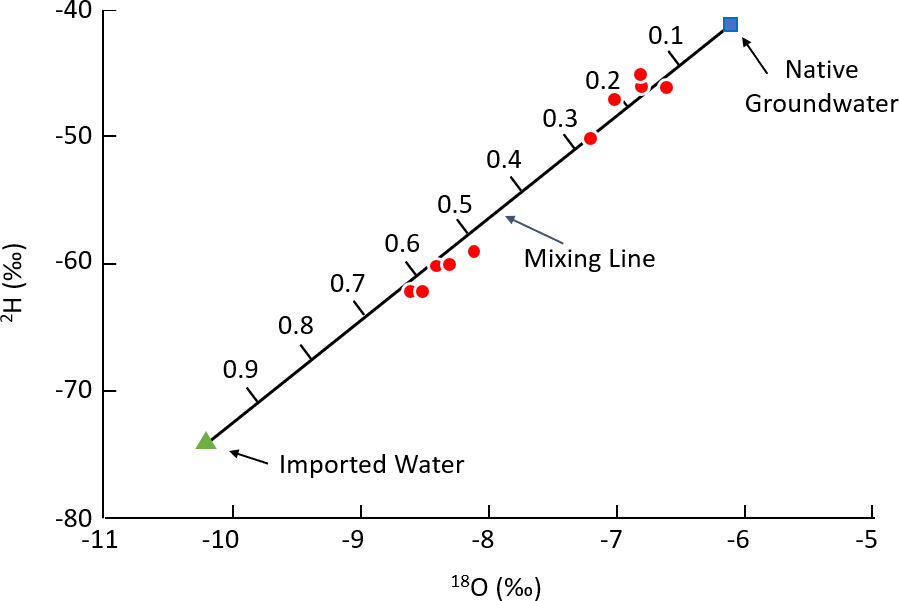
A good example of the use of environmental tracers to determine mixing fractions is provided by a study of artificial recharge in the Santa Clara Valley, California (Figure 12; Muir and Coplen, 1981; Coplen et al., 2000). Groundwater use in the Santa Clara Valley led to groundwater depletion, and an aqueduct was created to import water from northern California. The imported water was discharged into streambeds and percolation ponds to artificially recharge the groundwater. A study subsequently took place to determine the spread of the imported water from the sites where it was introduced, and its contribution to groundwater pumped by downstream wells. The δ 2H and δ 18O composition of native groundwater was determined to be -41 ‰ and -6.1 ‰, respectively, based on groundwater samples collected in areas unaffected by imported water. The mean value of imported water was -74 ‰ and -10.2 ‰ for δ 2H and δ 18O, respectively. Groundwater samples downstream of the artificial recharge sites ranged between -45 and -62 ‰ for δ 2H and -6.6 and -8.6 ‰ for δ 18O, and thus were intermediate between the native groundwater and imported water values (Figure 13). The authors used Equation 8 to estimate the contribution of imported water to downstream wells at between 10 and 70 percent. Lower fractions of imported water were estimated for wells furthest from the artificial recharge sites, with fractions between 10 and 20 percent recorded for wells up to 4 – 5 km downgradient.