6 Exercises
Exercise 1
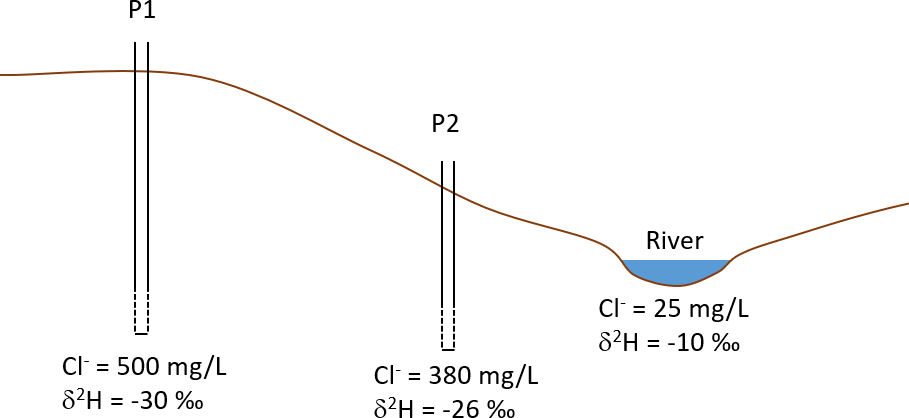
Water samples are collected from two piezometers and from a river. The piezometer that is closest to the river (P2) is believed to be a mixture of groundwater flowing towards the river (represented by piezometer P1), and river water that moves into the aquifer during high river flows. Assuming that the water obtained from piezometer P2 is a mixture of water from piezometer P1 and from the river, calculate the relative proportions of the two sources in the mixed sample from P2. Use Equation 7 and calculate the mixing proportions independently using chloride and deuterium.
Click here for solution to exercise 1
Exercise 2
Groundwater samples were collected from two wells in a confined aquifer. The wells are located along a flow line and are 13 km apart. The 14C activities on groundwater from these wells were determined to be 79 and 56 pmC. Assuming an initial 14C activity of 90 pmC at the time of groundwater recharge and that there are no chemical reactions affecting 14C activities, calculate the horizontal groundwater velocity. (The half-life of 14C is t1/2 = 5730 years, and its decay constant is 1.21 × 10–4 per year.)
If the head difference between the two wells is 2 m, what value of hydraulic conductivity would be consistent with the tracer data. Assume an effective porosity, ne, of 0.2.
Click here for solution to exercise 2
Exercise 3
A piezometer nest in an unconfined aquifer has a short screen which is 10 m below the water table. The CFC-12 concentration measured on groundwater obtained from this piezometer was 93 pg/kg. Based on a salinity of 400 mg/L, recharge temperature of 21 °C, and atmospheric pressure at recharge of 0.917 atm, the atmospheric concentration at the time of recharge is calculated to have been 225 parts per trillion by volume (pptv). This corresponds to the atmospheric concentrations in 1976 (Figure 6). If sampling took place in 2015, this reflects a water age of 39 years. Assuming an age of zero at the water table, a porosity of 0.2 and an aquifer thickness of 50 m, use Equation 10, to calculate the aquifer recharge rate.
Click here for solution to exercise 3
Exercise 4
A water sample extracted from an irrigation well had a 3H concentration of 10 TU. The 3He/4He ratio of dissolved helium was also measured, and after correcting for helium due to other sources, the 3He concentration due to decay of 3H (termed the tritiogenic 3He concentration) was estimated to be 5 TU. Calculate the apparent age of the water sample using Equation 3. (The decay constant for 3H is λ = 0.0558 y-1.)
