7.3 Groundwater as a Waste Repository (Out of Sight, Out of Mind)
On much of the Earth where there is some form of human activity generating chemical constituents that enter the subsurface causing changes in groundwater chemistry. Millions of different chemical compounds have been produced around the globe and many more are developed each year. As a result, many different chemical compounds have leaked into the subsurface in the past 100 years but only several thousand of these are currently known to cause detectable groundwater contamination. Figure 61 displays the types of chemical constituents that cause degradation of groundwater quality. Chemical constituents originating from human activities are known as anthropogenic compounds to distinguish them from the natural elements (geogenic elements) in groundwater. Most changes in groundwater chemistry are not detrimental to humans or ecological systems; however, those that can harm human health and the environment have become immense problems in many regions. The first recorded discovery that groundwater contamination can cause much human illness and death was made by a physician, John Snow, who in 1854 traced the cause of a cholera epidemic in London, England to a water well that was contaminated with human sewage. Although groundwater has been more protected since that time and causes fewer epidemics, illness and death due to groundwater contamination is widespread around the globe.

Figure 62 shows many of the activities and sources of chemicals in cities and rural areas that commonly cause groundwater contamination. In addition to the contaminants shown in Figure 62, urban areas have networks of sewers which often leak at joints and breaks. Suburban and rural areas have septic tanks that contribute to groundwater contamination. Each of the categories of contamination shown in Figure 62 results in a zone of groundwater contamination that extends away from the source in the direction of groundwater flow and, owing to its shape, it is known as a plume. Like the natural constituents in groundwater, the anthropogenic compounds migrate from recharge areas to discharge areas or to water wells. Some plumes travel to their discharge areas within a few years or decades, while others travel much longer paths with groundwater residence times of centuries or more. These longer and deeper flow paths are illustrated in Figure 35 and Figure 38. Some plumes disappear due to natural degradation or assimilation by subsurface chemical and/or microbial processes, or by dilution. However, contaminants in other plumes may not be removed or may be converted by chemical or microbiological processes into other contaminants that persist and continue migrating along the flow path until they are discharged.

Before the Industrial Revolution two centuries ago, the chemical constituents from human activities entering the subsurface were mostly natural nitrogen and organic compounds from humans and the waste of their animals. These chemicals rarely caused harm because they were readily assimilated or degraded by natural processes. In general, the subsurface has a huge capacity for assimilation (ability to clean itself) of many anthropogenic chemicals, which provides protection from most of the chemicals produced in our modern industrial society. However, in recent time we have released sufficient anthropogenic chemicals to overload the subsurface system to the point where the assimilative capacity for these compounds has been exceeded. Beginning during the Industrial Revolution of the early nineteenth century, with its capability to extract useful substances from coal and other geologic materials, new varieties of compounds were released into groundwater, including metals and coal tars. These were of limited societal concern at the time owing to low population density, limited use of groundwater, and sparse industrial activities.
Groundwater contamination sufficient to cause concern began in Europe and North America in the 1950s when:
- increased industrial production created the consumer society and the ubiquitous releases of chemicals to the subsurface;
- municipalities started spreading sewage sludge on land and using dumps for solid and liquid wastes, which evolved into engineered landfills;
- the “green revolution” led to the development and expansion of machine‑dependent agriculture with chemical fertilizers, herbicides and pesticides;
- industries began manufacturing complex organic chemicals for use in cleaning, fire protection, personal care products, pharmaceuticals, and other applications, which became used ubiquitously, some being released directly to the environment (e.g. during firefighting) and others present in wastewater, which commonly leaks from sewers or is released directly to the subsurface via septic tank leach fields; and,
- use of groundwater increased to avoid using water from polluted rivers and lakes.
Most of the millions of manufactured organic chemicals are either not mobile in groundwater because they bind to mineral surfaces or are quickly degraded by natural bacteria and have little impact. Some, however, now known to total a few thousand, are mobile and degrade slowly if at all (are refractory), or are converted by natural processes to other more harmful contaminants. Many of these contaminants can be conveyed by groundwater flow and, in most industrialized countries, there have been 50 to 70 years of migration of mobile refractory contaminants in groundwater, leading to long plumes (100s to 1000s of meters in length).
An important step in assessing whether or not a pumped aquifer is likely to show anthropogenic contaminants, that may pose harm to groundwater users, is the presence of detectable tritium. Modern groundwater is identified by the presence of detectable tritium (radioactive water molecules) at extremely low concentrations. Tritium was released to the atmosphere beginning in the 1950s and more so in the early 1960s from above ground nuclear testing and incorporated into precipitation that recharged groundwater. In most countries that have developed groundwater, nearly all of the important aquifers show the presence of atmospheric tritium, indicating at least some of the groundwater molecules recharged the aquifers during the past several decades. For example, 3,493 groundwater samples in the United States Geological Survey database have been analyzed for tritium and 2,702 were positive (77%). Modern groundwater is likely to contain other mobile anthropogenic constituents as well. Generally, more and more types of chemical compounds are found in groundwater, but the long-term consequences of this trend on human health are not known, especially with regard to the impacts of mixtures of different types of contaminants.
One of the features that sets groundwater apart from surface waters is that contamination of groundwater has a much longer residence time. If we were able to “wave a wand” and cease all further releases of contaminants into groundwater, the contaminants already in groundwater would eventually be flushed out or completely assimilated because most plumes are part of dynamic groundwater flow systems with assimilation capacity. But for many plumes, the time required for flushing or assimilation will be centuries or millennia. One example is the fumigant dibromochloropropane (DBCP), whose use in farming areas in the U.S. was banned in the 1979, yet DBCP was still widely detected in California groundwater as late as 2017, 38 years after its use was stopped. In contrast, flushing and attenuation in rivers and lakes typically requires only weeks or months after inputs cease. Therefore, given that groundwater contamination can persist for long periods of time, an important goal is to prevent more contamination from entering groundwater so that contaminants do not start the long groundwater journey.
Much of the reason that groundwater has become degraded is because humans often have an “out of sight‑out of mind” perspective. However, even when groundwater was on our minds, there were errors in our assumptions about groundwater and thus unrealistic expectations of what would happen when wastes were released into the subsurface. For example, consider on‑site sewage treatment and disposal systems (septic systems) that are used to dispose of household sewage. Septic systems are wastewater‑treatment installations that are emplaced underground. Typically, a septic system has a tank and a drain field with horizontal perforated piping for the wastewater to infiltrate through the vadose zone into groundwater (Figure 63). While septic systems provide the first line of defense against the spread of disease because they generally remove pathogens (bacteria and viruses), they are not designed to remove the nitrogen and many other contaminant types derived from household chemicals and pharmaceuticals. Figure 63 shows a septic system and its plume in the groundwater shortly after the septic system was installed. With time, the plume of refractory compounds becomes longer and longer until it reaches a receptor such as a well, river, lake or ocean estuary.

About one‑fifth of households in the United States, Canada and the European Union have (on‑site) septic systems. Septic plumes contain nitrate and phosphorus, as well as chemicals from pharmaceuticals, personal care products and food additives. In the 1950s, septic systems became a standard household feature of many areas in the rapidly developing suburban areas of the United States. This was based on engineering analysis, but the analysis assumed a strong degree of plume attenuation by strong assimilative capacity of the subsurface. The septic plumes were not expected to extend far from each septic system and the occurrence of persistent nitrate, phosphate, pharmaceuticals and food additives in plumes was not anticipated. In fact, most households with septic systems also have a water well on the property (Figure 63) and often not far enough removed from the septic systems to avoid capture of chemicals in the plume from the septic leach field. Overall, the magnitude of impacts of septic system contaminants in groundwater on human health are poorly understood, but what is not poorly understood is that septic system contamination is damaging the water quality and ecology of many lakes, rivers, estuaries and bays because the discharge of nitrogen and phosphorus to these water bodies causes eutrophication. These effects are also caused by plumes from leaky city sewer systems.
Likely the main problem for human health from human and animal sewage going into groundwater is contamination by pathogens, for example some strains of Escherichia coli (E. coli) bacteria and viruses. Bacteria are generally not mobile in sand aquifers because they are large enough to be filtered out by the sand, but can be mobile in gravel aquifers, karst and fractured rock aquifers, and as viruses are much smaller, they are more mobile than bacteria in aquifers. Harmful viruses can remain active for as long as three years and may travel many kilometers in high permeability aquifers. E. coli are bacteria that are easy to detect in water samples and their presence is an indicator of the potential presence of viruses. Although there are now effective methods for sampling and analysis of viruses in well water, such testing is rarely done, so that the frequency of illness attributable to viruses in groundwater is poorly known. E. coli are commonly found in household wells.
The so‑called green revolution in food production is another global example of how new science was to bring large benefits, but inadvertently brought immense adverse groundwater side effects. This started in the 1940s with large changes in crop genetics (plant cross breeding), but that aspect of the revolution soon ended when agriculture became highly mechanized and dependent on manufactured fertilizers, herbicides, and pesticides. A significant amount of the nitrogen in the fertilizer ends up in groundwater as nitrate where it is harmful to human health and detrimental to ecosystems. As a result of chemical agriculture and sewage leaching into groundwater, nitrate is now the most common contaminant in groundwater around the globe.
When charting the history of groundwater contamination over the past 70 years, one finds that the development and production of new chemical products often has unintended negative consequences for groundwater quality (Figure 64).

The most recent example of a scientific advance that was designed to improve everyday life having a negative impact on groundwater is the development of several thousand fluorinated organic chemicals known collectively as PFAS (per‑ and poly‑fluoroalkyl substances) as shown in Figure 65. These are essential to producing modern commercial products such as Teflon, Gore‑Tex, fire‑fighting foams and flame‑retardant clothes and bedding. They are widespread globally in groundwater, where they are mobile and persistent. They bioaccumulate (accumulate in tissues of organisms exposed to them) so now most human beings and many other animals on Earth have PFAS in their blood and cells. Accumulation of PFAS in humans and animals has been related to problems such as disruption of the immune and metabolic systems, complications in neurodevelopment and cancer (Sunderland et al., 2019).

Another scientific advance that may negatively impact groundwater is the development of nanoparticles which are used in the manufacture of many products including scratchproof eyeglasses, crack‑resistant paints, anti‑graffiti coatings for walls, transparent sunscreens, stain‑repellent fabrics, self‑cleaning windows and ceramic coatings for solar cells. Nanoparticles are in our waste and will likely be found in groundwater. If nanoparticles are found to be common in groundwater, we do not know what the consequences will be.
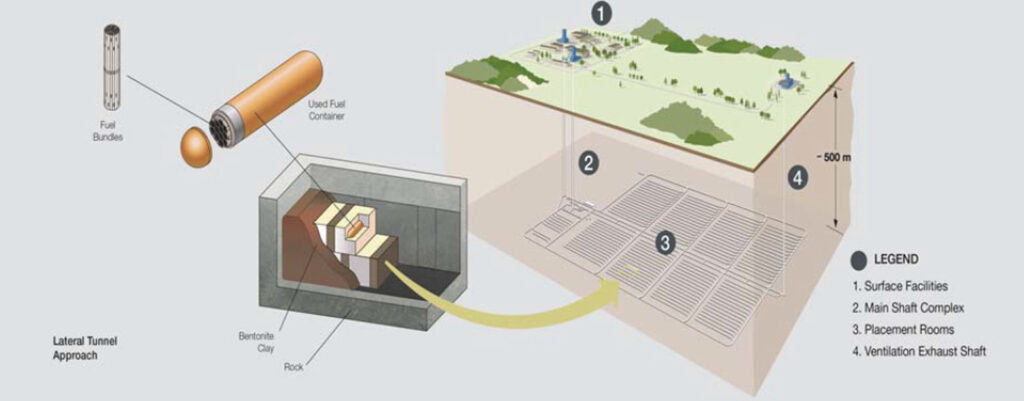
In this discussion of groundwater contamination, the outcomes have all been negative, but there is a concept for using the groundwater zone as a waste repository aimed at positive outcomes. This is the development of deep geological repositories (DGR) for high‑level nuclear waste. This waste needs to be contained for a million years. Therefore, the objective is to entomb the waste in deep zones where the radioactivity will be contained on a geological time scale. In other words, store the waste where the groundwater is not part of the active hydrologic cycle under current conditions. Many countries are designing and some are operating DGRs in very low permeability geologic layer sat depths on the order of 450‑600 meters below ground surface where the groundwater is saline, stagnant and shows evidence of having resided in these layers for millions of years (Figure 66). There are many other types of hazardous wastes with chemicals that are not radioactive but degrade slowly and DGRs are also an option for isolating some of these wastes from the environment.