Natural Chemical Constituents in Groundwater
Natural waters contain dissolved elements known collectively as Total Dissolved Solids (TDS), nearly all of which are natural inorganic salts. Water with less than 1000 milligrams per liter (mg/L) of TDS is considered fresh, while water with more TDS (>4000 mg/L) is classed as brackish, water with higher TDS (>10,000 mg/L) is saline (sea water ~ 35,000 mg/L), and brine (>35,000 mg/L) is extremely salty. Table 1 summarizes the terms used to describe the TDS levels of water. When a well is drilled just about anywhere on the globe, the shallow groundwater has low TDS (is “fresh”) and is good for drinking if it is not excessively impacted by chemicals from human activities. At greater depth, the water is brackish, deeper yet it is saline and below this, the water is typically brine. This change of water with depth from fresh to brackish to saline to brine is nearly universal, except in a few places where there is no fresh or brackish water at any depth because all the groundwater is salty.
Table 1 –Terms for varying degree of dissolved solids in water.
| Total Dissolved Solids (milligrams/liter) | Water Description |
| < 1,000 | fresh |
| 1,000 – 4,000 | lower end of this may be drinkable |
| > 4,000 – < 10,000 | brackish |
| > 10,000 – < 35,000 | saline |
| 35,000 | sea water |
| > 35,000 | brine |
Natural fresh groundwater contains dissolved elements in three categories: major constituents; minor constituents; and, trace constituents. The amounts of major, minor, and trace elements in the groundwater depend on the climate in the recharge areas, the chemical conditions of the vadose zone and the geology of the groundwater system through which the water flows. It is useful to know the types of elements that might be present in groundwater because it is not feasible to measure every element of the periodic table in a water sample. Typically, only 8 or 9 elements constitute over 95% of the mass of dissolved solids in groundwater. Regardless of the setting, some elements are so pervasive that they are always part of a basic chemical analysis of groundwater.
TDS in groundwater is mostly in ionic form (i.e., positively or negatively charged ions). H+ and OH– ions are always present in water because water molecules, H2O, dissociate into H+ and OH–. This seemingly simple phenomenon is actually fascinating and complex, although only some basic concepts and terminology are discussed here. The amount of H+ ions in a particular water sample is usually expressed as pH. As you may know, a pH of 7 in pure water is called neutral, which is a condition in which the H+ and OH– ions are balanced. The pH of natural groundwater varies. Figure 50 lists the variation of pH as 6 to 9, with 7 to 8 a more typical range. For a pH of 7 in pure water there are 0.0001 mg/L of H+ ions. Although present at very low concentrations compared to the other species, H+ and OH– are very important to a multitude of reactions that occur in groundwater, including weathering, dissolution, precipitation, and microbiological and abiotic transformations.
Many chemical constituents are dissolved in groundwater as illustrated in Figure 50. The six constituents typically present at highest concentrations in groundwater are known as major ions: including positively charged ions called cations (sodium, Na+; calcium, Ca2+; magnesium, Mg2+) and negatively charged ions called anions (bicarbonate, HCO3–; sulfate, SO42-; chloride, Cl–). These usually make up nearly all the mass in the measured TDS value. The minor constituents that may be present include: potassium, K+; manganese, Mn2+; iron, Fe2+; silicon, Si4+; nitrate, NO3–; fluoride, F–; and dissolved organic carbon, DOC. These minor constituents, generally have a concentration in the range of 0.1 to 5 mg/L. Many trace elements are present at lower concentrations, sometimes below our ability to detect them with commonly used analysis methods.
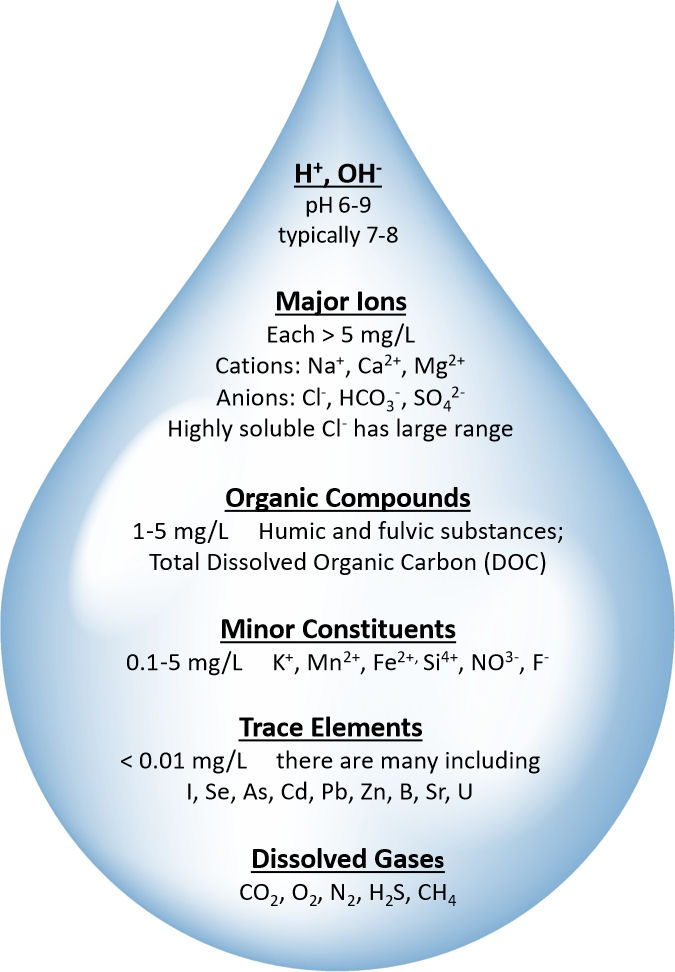
The amounts and types of constituents in these major, minor and trace categories give water the chemical portion of its natural “quality”. Other components of water quality are temperature, color, odor, turbidity and microbial content. Some of the natural constituents in groundwater can be harmful at higher concentrations, but contribute to good health when at lows concentrations (e.g., iodine, fluoride, and selenium).
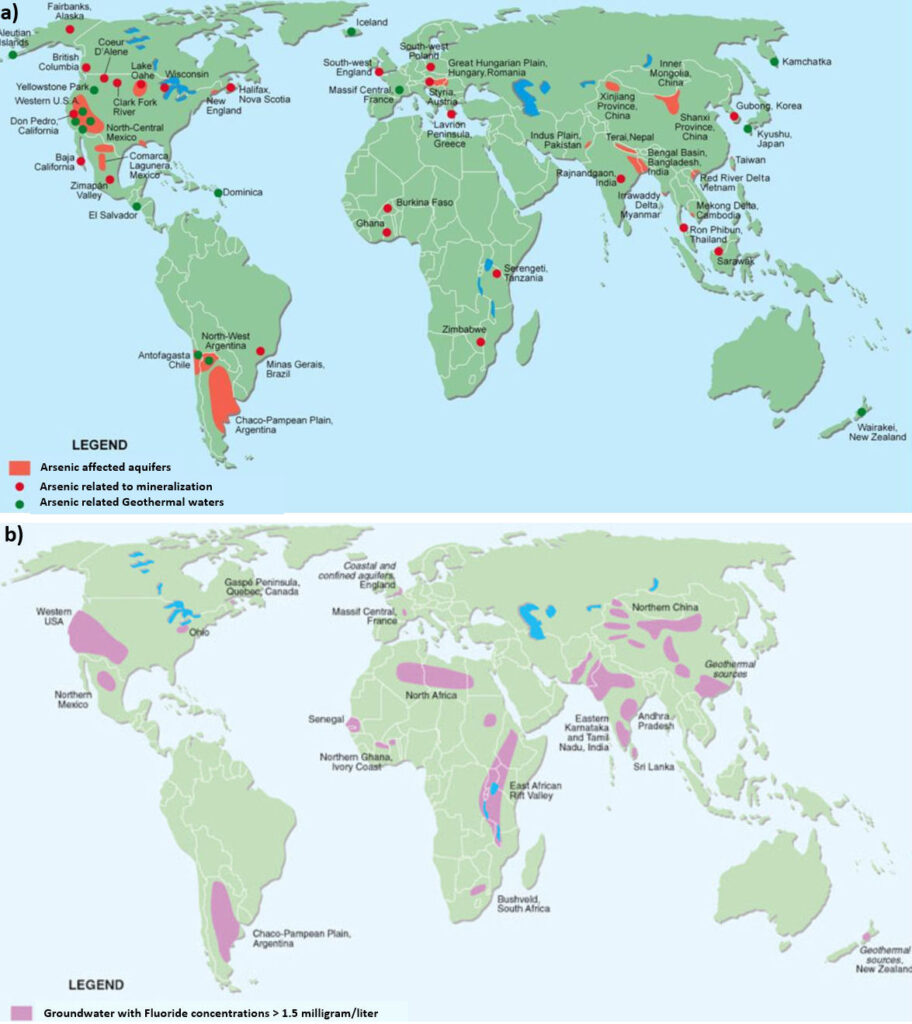
Nearly all fresh groundwater that has no significant contamination from human activities is safe to drink but there are important exceptions, most notably where natural groundwater contains excessive amounts of arsenic or fluoride or in Karst systems. Drinking water with excessive arsenic causes many health problems including cancer. Arsenic occurs naturally at harmful levels in groundwater in more than 60 countries, affecting more than 150 million people. While low levels of fluoride (on the order of 1 to 2 mg/L) have been shown to reduce the occurrence of dental cavities, excessive fluoride causes dental fluorosis, or worse, crippling skeletal fluorosis. It is estimated that more than 200 million people consume water with high fluoride concentrations including people in China, India, United States, Canada, and parts of Africa and South America.
Although safe levels have been established for many other naturally occurring elements, such as cadmium, mercury, lead and silver, these rarely occur at harmful concentrations in natural groundwater. The occurrence of harmful levels of arsenic and fluoride results from a combination of two factors: the presence of arsenic‑ or fluoride‑rich minerals in geologic materials and chemical conditions that facilitate their dissolution and release into groundwater.
In countries relying heavily on domestic wells for supply such as Bangladesh, arsenic may cause harm. If a well is drilled in a geologic formation where arsenic‑bearing minerals are present and oxygen is absent, dissolved arsenic and iron will be present in the extracted groundwater. Water in shallow wells typically has enough dissolved oxygen that iron minerals precipitate, removing arsenic from the water. However, other health hazards may be of concern for shallow hand‑dug and poorly constructed drilled wells, such as bacterial contamination from surface runoff.
Arsenic and fluoride in groundwater have caused and continue to cause severe health effects in many rural areas where poverty prevents avoidance. But they also threaten health in wealthier areas where millions of private domestic well owners consume groundwater rich in arsenic or fluoride without awareness because private wells are not included in government testing and regulation. The locales known to have excessive amounts naturally occurring arsenic and fluoride are shown in Figure 51a and b.