9.2 Aquifer Lithology and Mineralogy
Although quite common, not all aquifers undergo a transition to Na-HCO3 type water. Detailed water chemistry of groundwater at several different depths within the Stripa granite, central Sweden, indicated an evolution from a dilute Ca-HCO3 type water to a deeper Na-Cl type water with elevated Ca and SO4. The F concentrations ranged from 0.22 mg/L in the shallow groundwater to 5.6 mg/L in the deep (> 900 m) groundwaters. The pH was typically 9 to 10 from 300 to 1000 m depth and the HCO3 alkalinity decreased to unusually low concentrations with depth (< 10 mg/L). Fluorite and apatite are the only F-bearing minerals that were identified and they occur primarily in fracture-fillings where water flow paths are important (i.e., carry significant volumes of flow). Fluoride concentrations do not correlate well with alkalinity or Cl (Figure 7). The dominant control of the higher F concentrations at depth seems to be the elevated pH concentration.

Figure 7 – a) Fluoride concentrations plotted against HCO3 concentrations for groundwaters in the Stripa granite over depths of 80-1000 m. b) Fluoride concentrations plotted against Cl concentrations for the same samples as in (a).
The F concentrations can be simulated by assuming calcite and fluorite solubilities have reached saturation but only if saturation index values (SIs) are set at oversaturation of about 0.8 for each mineral. The resultant pH from the simulation is also within 0.1 of the measured value. This amount of supersaturation is consistent with calculated values for the numerous other deep samples in the data set. Figure 8 shows the SI values for calcite and fluorite plotted against Cl concentrations.
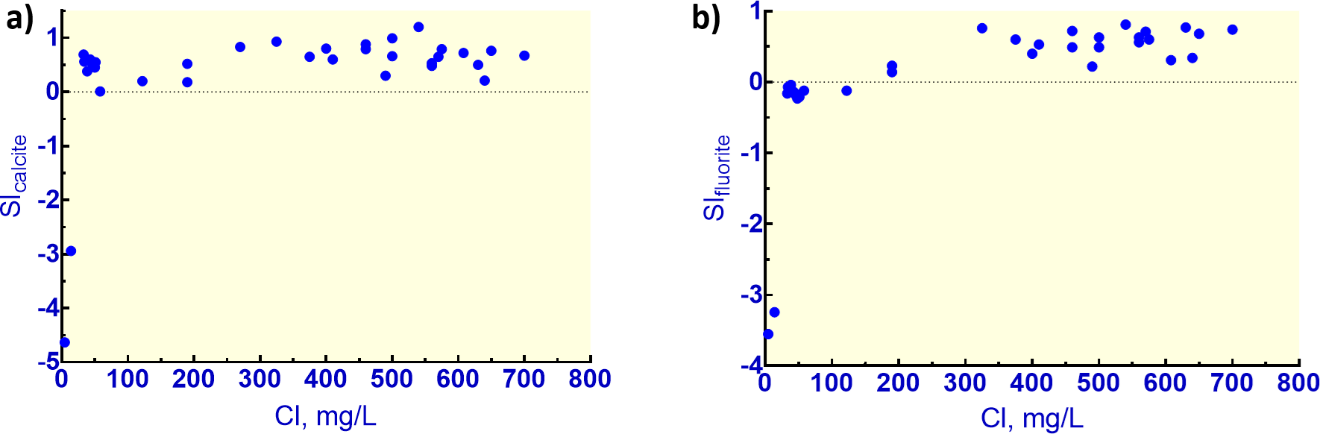
Figure 8 – a) Calcite saturation indices with Cl concentration which roughly corresponds with depth for Stripa granite groundwaters (Nordstrom et al., 1989). b) Fluorite saturation indices for the same samples shown in (a).
Similar water compositions were encountered in the Toki granite at depths of 200 m to 1000 m at the Mizunami underground research laboratory. In the Miocene/Pliocene/Pleistocene sediments, groundwaters were generally Ca-HCO3 to Na-HCO3 grading into Na-(Ca)-Cl type waters with depth, especially in the granite (Iwatsuki et al., 2005). Concentrations of F varied from 0.1 mg/L in the shallow dilute waters to 15 mg/L in the deeper, higher Cl waters.
The F concentrations are limited by fluorite solubility equilibrium at higher pH values, higher F concentrations and generally occur at depths greater than 75 m as reflected in the saturation indices which are at or above saturation (Figure 9).

Figure 9 – a) Calcite, and b) Fluorite saturation indices with F concentration and varying depth. F concentration and depth based on data from the Mizunami Underground Research Laboratory (Iwatsuki et al., 2005).
Reactions and processes that lead to a decrease in Ca concentrations, usually by calcite precipitation, will cause F concentrations to increase if a fluorinated calcium mineral is present in the aquifer.
