2 History of Fluoride in Water and the Recognition of Fluorosis
The chemical element fluorine was thought to be an element in compounds such as fluorspar, a common name for fluorite, from the beginning of the 19th century and had been detected in bones, teeth, and ivory (Emsley, 2001; Roholm, 1937). At the suggestion of André-Marie Ampere, Sir Humphrey Davy named it fluorine in 1813 before it was finally isolated in 1886 by Moissan.
Elemental F exists as a diatomic gas, F2. When dissolved in water it has a strong affinity for one electron to attach to its outer valence shell and becomes a fluoride ion, F–. Methods for analytically determining F in water were available before 1886 but were not always reliable. Gooch and Whitfield (1888) attempted to determine F in Yellowstone’s thermal waters but could not detect it. Allen and Day (1935) also attempted to determine F in Yellowstone’s waters and discovered a flaw with Gooch and Whitfield’s procedure. Using an improved method, they found 15 to 20 ppm F in alkaline thermal waters. Indeed, quantitative methods for determining F in water continued to improve during the 20th century in parallel with the discovery of dental and skeletal fluorosis and their association with high-F groundwaters.
In 1888, German dentist Kühns reported on the occurrence of discolored teeth (brown and black) among residents in Durango, Mexico, who had been drinking from a hot spring. He suspected iron and/or manganese staining, but it was most likely fluoride. In 1901, Dr. Frederick McKay arrived in Colorado Springs, Colorado, and opened a dental practice having just graduated from dental school. He discovered that many residents had seriously browned and mottled teeth (“Colorado brown stain”) yet negligible signs of tooth decay. Through his perseverance over 30 years, he discovered the cause was the ingestion of high-F drinking water. There are numerous fluoride minerals that occur in the Pikes Peak area around Colorado Springs including the relatively rare mineral cryolite, Na3AlF6, which is fairly soluble (Roberson and Hem, 1968). Undoubtedly, the abundance of these minerals contributed substantially to the F-rich drinking water supplies of Colorado Springs residents. Dr. McKay continued to seek the cause of mottled teeth enlisting the help of dental experts and published several papers on their research. They were convinced that it had to be something in the water supply but could not identify the substance until many years later.
The first experimentally demonstrated relation between high-F dosages and dental defects in experimental rats was reported in 1925 (McCollum et al., 1925) and subsequently confirmed by other researchers. However, there was not thought to be the same effect for human teeth. It was not until 1931 when chemists working for ALCOA in Bauxite, Arkansas, discovered that the occurrence of mottled teeth in some residents was caused by fluoride in the groundwater (derived from using cryolite as a flux in the electrolytic extraction of aluminum). Dr. Churchill, chief chemist for ALCOA, contacted Dr. McKay and suggested he send out water samples from Colorado Springs for F determinations. These results and others linking high-F drinking water in five states to mottled teeth were published in 1931 (Churchill, 1931). Further confirmation came the same year when researchers at the University of Arizona Agricultural Experiment Station showed that a high-F water fed to white rats produced defects nearly identical to those in the community of St. David, where residents were using well water with up to 7 ppm F (Smith et al., 1931).
Independently and concurrently with the work of McKay and others, Henri Velu was investigating the cause of animal and human teeth disorders in North Africa during the early 1920s. He concluded that high phosphate zones in rocks and sediments were mobilizing fluoride and causing dental fluorosis (Roholm, 1937; Velu, 1931).
The first major task for H. Trendley Dean when he was appointed head of the Dental Research Section of the newly formed (1931) National Institute of Health was to determine whether F was the sole cause of mottled teeth and secondly to determine if there is a threshold concentration below which mottling is not observed. Based on a considerable amount of data collected across the United States, he developed the clear relationship shown in Figure 1. The slim margin between healthy children’s teeth and mottled teeth was estimated to be a concentration of 1 mg/L (Dean, 1936). This concentration has found substantial support by numerous subsequent studies (e.g., as indicated by references in Ali et al., 2016; Jagtap et al., 2012; NRC, 2006).
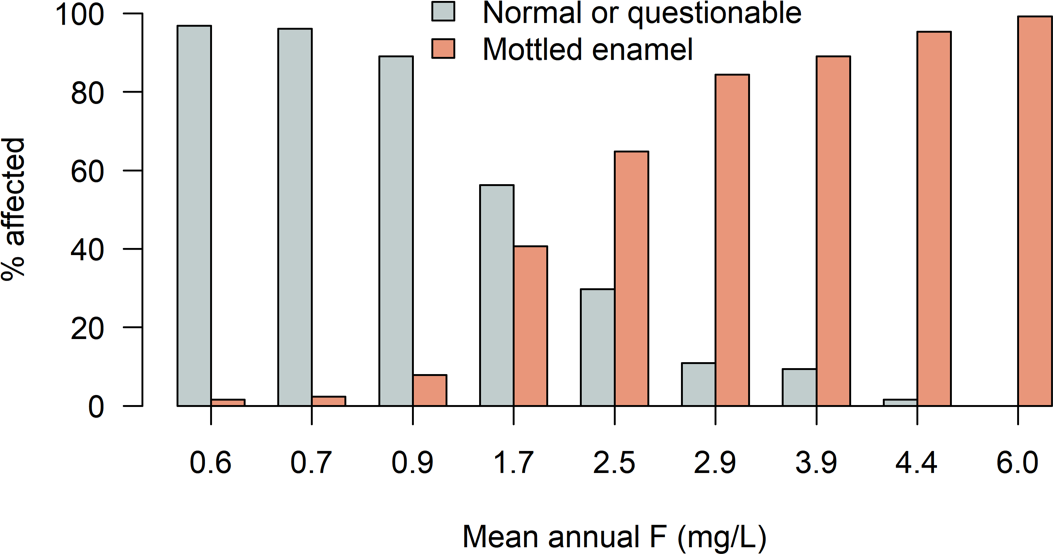
Figure 1 – Prevalence of mottled teeth in areas as it relates to the mean annual fluoride concentration in the water supply (modified from Dean, 1936).
Dean made observations on 7,257 children aged 12 to 14 in 21 cities using a 5-point classification system. This work led to further long-term trials with fluoridation in several cities and in 1962 the United States Public Health Service provided a drinking water standard for F of 0.7 to 1.2 mg/L depending on mean annual temperature. Following the passing of the National Environmental Protection Act in 1970 and the formation of the United States Environmental Protection Agency (EPA), a national interim drinking water standard of 1.4 to 2.4 mg/L was proposed in 1975. In 1986 the EPA established a maximum contaminant level goal (MCLG, non-enforceable) of 4 mg/L for the prevention of skeletal fluorosis and the maximum contaminant level (MCL, enforceable but interim standard) was and is the same. A secondary MCL (SMCL, non-enforceable) of 2 mg/L was also recommended for cosmetic reasons. The National Academy of Sciences reviewed these findings from 2002 to 2006 and recommended that the 4 mg/L MCLG is too high and that the EPA should establish a lower standard (NRC, 2006). In 2015 the United States Public Health Service recommended lowering the F concentration for fluoridation of municipal water supplies to 0.7 mg/L based on a study by Heller and others (2007). Details of the debates, the toxicological and epidemiological evidence, and known exposures in the USA are provided in NRC (2006), federal records, and Freeze and Lehr (2009).
During the same time period that dental fluorosis was being identified and studied in the USA, notable worldwide examples were reported. Mottled teeth were reported in China by 1930 (Anderson and Stevenson, 1930); fluorosis was recognized in India by 1937 (Day, 1940; Shortt et al., 1937a; Shortt et al., 1937b; Wilson, 1939b); in Japan by 1931 (Williamson, 1953); in England by 1933 (Ainsworth, 1933; Murray, 1996a; Wilson, 1939); and in Africa mottled teeth in mammals were recognized by 1922 (Velu, 1922) and in people by 1931 (Ockerse, 1953; Tekle-Haimanot et al., 1987; Velu, 1931). In Morocco and Algeria, the condition observed in animals was referred to in French as “le Darmous” (Velu, 1931).
In the United Kingdom, the earliest observations of mottled teeth were recorded among school children in Essex, where an association was established with fluoride in local Chalk groundwater at concentrations of 4.5 to 5.5 mg/L (Ainsworth, 1933; Ainsworth, 1934; Hoather, 1953). By contrast, during World War II, the dental health of children evacuated from the town of South Shields in north-east England was noted to be much better than that from the children of the host county in the north-west, a factor attributed to the South Shields water supply having a fluoride concentration of 1.4 mg/L (Mullen, 2005).
The Danish toxicologist Kaj Roholm published a definitive book on F toxicity that became the standard for many years, as a result of his investigations into the symptoms of workers at a chemical plant in Copenhagen exposed to cryolite (Roholm, 1937).
