9.1 Na-HCO3 Groundwaters and the Aquia Aquifer, Maryland, USA
The recognition of groundwaters evolving from a Ca-HCO3 type water in recharge areas to a Na-HCO3 water downgradient has been documented since at least the 1930s (Cederstrom, 1946; Foster, 1937; Foster, 1942; Foster, 1950) for the Virginia Atlantic coastal plain and is standard material in groundwater geochemistry textbooks (Appelo and Postma, 2005; Drever, 1988). Fluorosis was also widespread among children in parts of the southern Virginia coastal plain since the 1930s (Cederstrom, 1939). This chemical evolution of groundwater is found worldwide in many different aquifers and hydrologic conditions such as Ethiopia (Bretzler et al., 2011), Mexico (Moran-Ramirez et al., 2016), Brazil (Gastmans et al., 2016), Mississippi delta (Borrok et al., 2018), Australia (Herzeg et al., 1991), and France (Huneau and Travi, 2008).
Geochemical modeling of groundwater-rock interactions frequently considers cation exchange to be a major process during the evolution of groundwaters where exchangeable clays are present (Postma et al., 2008). The basic concept is that recharge waters pick up Ca, CO2 and HCO3 from the soil zone and shallow aquifers to produce dilute Ca-HCO3 type waters in recharge areas and evolve to Na-HCO3 type waters from cation exchange with marine, Na-saturated clays that are downgradient. This process is often accompanied by sulfate reduction and organic matter decomposition which adds DIC to the water and promotes calcite precipitation (Back, 1966; Foster, 1950). Geochemical modeling of the Aquia aquifer in the eastern USA demonstrated this process utilizing a version of the PHREEQC code to explain the major changes in water composition with distance (Appelo, 1994) as shown in Figure 6. Similar modeling has demonstrated this same overall process for the Triassic East Midlands aquifer, United Kingdom, the Miocene Valréas aquifer, France, and the Cretaceous Aveiro aquifer, Portugal (Postma et al., 2008).
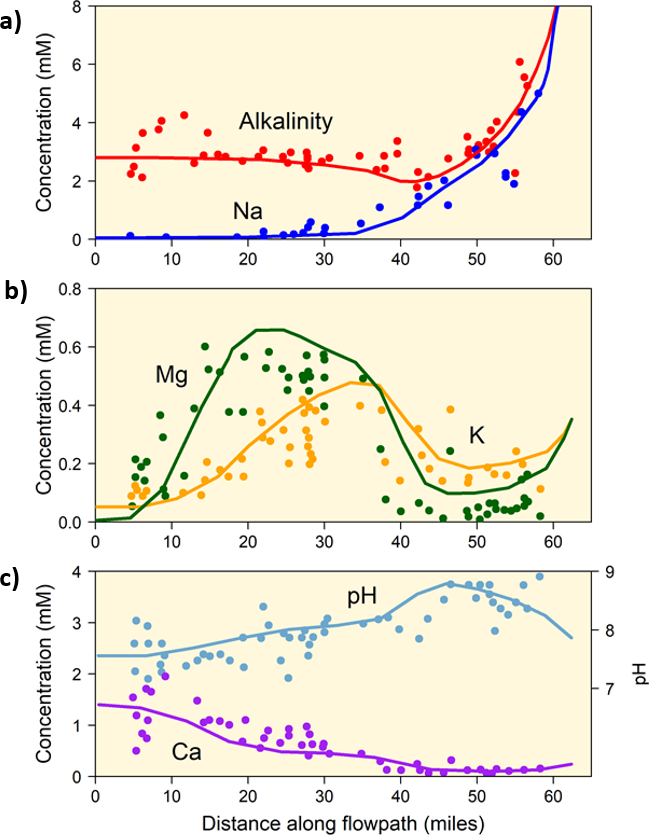
Figure 6 – Changes in a) Na, alkalinity, b) K, Mg, and c) Ca concentrations and pH with downstream distance (in miles, 60 miles is about 97 kilometers) in the Aquia aquifer, Maryland, USA. Data points are from Chapelle and Knobel (1983) and lines are simulations (from Appelo, 1994) based on 1-dimensional reactive transport with ion exchange acting chromatographically.
Previous studies have shown that the primary exchangeable clay in the Aquia aquifer was glauconite (Chapelle and Knobel, 1983) and the association of phosphorite with glauconite has long been recognized (Collet, 1908; Nathan, 1984; Notholt, 1980). Hence, areas where shallow marine sediments include glauconite and phosphorite, which later undergo freshening, will experience this transition from Ca-HCO3 type water to Na-HCO3 type water with a consequential rise in F concentrations (Cederstrom, 1946). In Cederstrom (1946), the highest concentration reported was 7.7 mg/L. Back (1966) compiled water-quality data for the Atlantic coastal plain and found F concentrations ranging from < 0.1 to 6.4 mg/L. McFarland (2010) has updated information on the hydrogeology and groundwater quality of this area and reported F concentrations as high as 18 mg/L with pore waters extracted from sediments as high as 30 mg/L. He also points to a phosphatic sedimentary source material combined with desorption of F from Fe oxyhydroxides. The basic pattern of cation exchange with calcite dissolution/precipitation was modeled as a chromatographic column with 1-dimensional reactive transport by Appelo (1994) and the comparison with field data was remarkably consistent (Figure 6). Calcite dissolved in the recharge zone and precipitated downgradient. Where sufficient data exist, this same pattern of increased F concentrations with the development of a Na-HCO3 water is often observed.
