9.3 Extremes of pH
Waters of low pH are highly reactive and have the capacity to dissolve minerals rapidly. If fluorite is present, the reaction produces high F concentrations. Mineralized areas may contain both pyrite and fluorite. Pyrite oxidizes to produce sulfuric acid which will dissolve fluorite readily. An example of such a condition occurs in the Red River Valley, New Mexico, where naturally acidic groundwaters from pyrite oxidation dissolve fluorite and produce elevated F concentrations. In one well, groundwaters in a debris fan were monitored for more than a year and had F concentrations of 8 to 11 mg/L with Ca concentrations of 300 to 400 mg/L. The pH varied between 3.5 and 3.7. Acidic waters such as these are accompanied by high Al concentrations because Al dissolves readily from aluminosilicate minerals and forms strong Al-F complexes. This complexing is reflected in Figure 10 in the trend line of increasing F with increasing Al concentrations for acidic samples. The lower the pH, the higher the Al and F concentrations in the acidic group with pH values less than 4.5. Even in much more dilute waters of moderate acidity (pH 5.5 to 6.5) the strong association of Al and F complexing is quite evident (Berger et al., 2015).
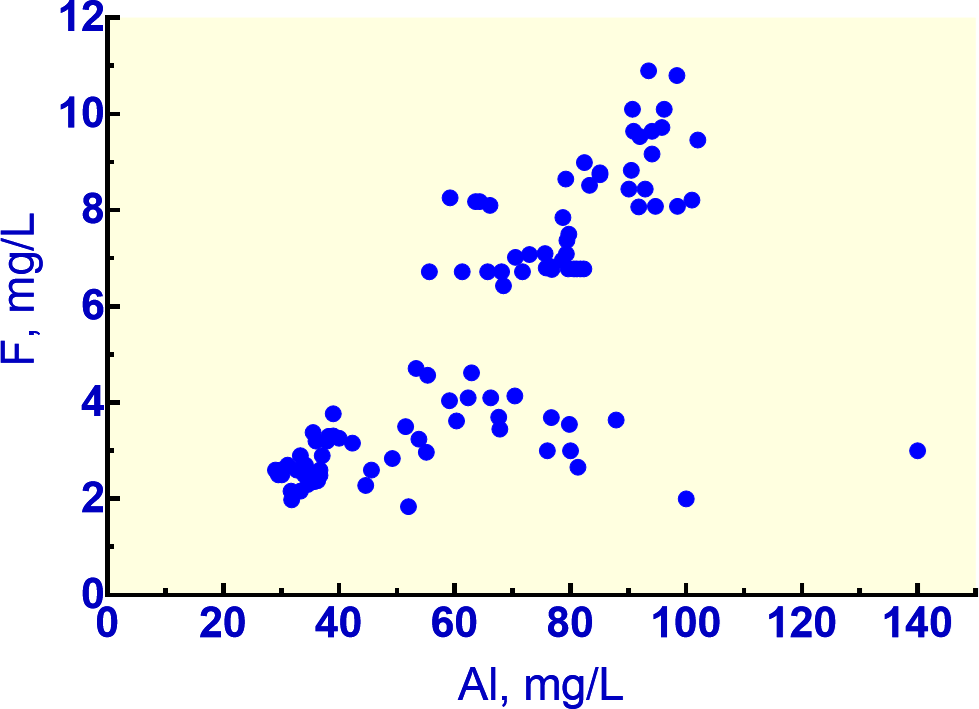
Figure 10 – Naturally acidic groundwaters (pH < 4.5) reflecting a strong association of Al with F as a result of Al-F complexing (data from Naus et al., 2005; Nordstrom et al., 2005).
The well-known Berkeley Pit next to Butte, Montana, has a pit lake with pH values from 2.17 to 4.15 and F concentrations ranging from 1.38 to 47.9 mg/L (Figure 11), based on data provided by the Montana Bureau of Mines and Geology. Although pit lakes could be classified as surface waters, they are often fed by groundwater and at the Berkeley Pit there are extensive underground mines connected to the base of the pit so that much of the acidity came from the flooded mines in addition to weathering of pit walls. The high F concentrations originated from subsurface fluorite in the mineralized zones. The waters with pH values above 3 were produced by lime neutralization of a portion of pit water which was returned to the pit after sludge removal. These data demonstrate that negligible fluoride is being removed even with lime addition.
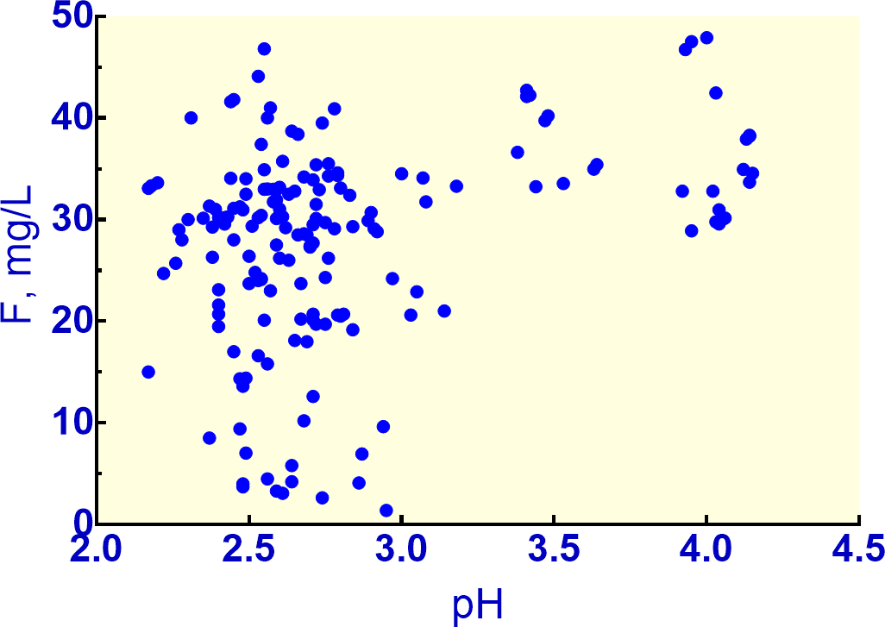
Figure 11 – Dissolved F concentrations plotted against pH for samples from the Berkeley Pit Lake, Butte, Montana, USA, during 1984-2019 (data provided by Montana Bureau of Mines and Geology).
There is not much that can limit fluoride concentrations at low pH. The solubility of fluorite is so high at pH values below 3 that it can fully dissolve in strong acid and produce toxic HF gas as shown in Equation 11 and Figure 12a. Fluorapatites are also highly soluble at low pH (Figure 12b).
| CaF2 + 2H+ → Ca2+ + 2HF (g) | (11) |

Figure 12 – a) Solubility of fluorite (in terms of F– concentration) as a function of pH (calculated and plotted from PhreeqcI using the phreeqc.dat database). At low pH, fluorite can completely dissolve. b) Solubility of fluorapatite (in terms of F– concentration) as a function of pH showing that it is even more soluble than fluorite at low pH but less soluble at pH values above 5.
In one of the mines in the Kola peninsula of northwestern Russia, Kraynov and others (1969) found 15,000 mg/L F in deep waters of the Lovozero massif. The pH of this water was about 12 with extremely high silica concentrations of up to 13,000 mg/L. The cause of the extraordinary F concentrations was the occurrence of the very soluble mineral villiaumite, NaF, along with fluorosilicate complexing.
