7.3 The Effect of Dissolved Inorganic Carbon (DIC) and Calcite Precipitation
If we take another 1:1 soluble electrolyte like NaHCO3 and increase its concentration to the fluorite solubility limit, we find that F concentration increases like it did when adding NaCl to the solution, except that the F concentrations are higher (Figure 5). If another component such as CO2 is added, then F concentration is higher still. When the concentration of NaHCO3 is at the same concentration as NaCl, the F concentrations is higher because a CaHCO3+ complex is formed and with more dissolved carbon from CO2, more of this complex is formed. Any complex formation requires more of the mineral to dissolve to reach equilibrium because the activity and concentration of the free ion has been decreased.
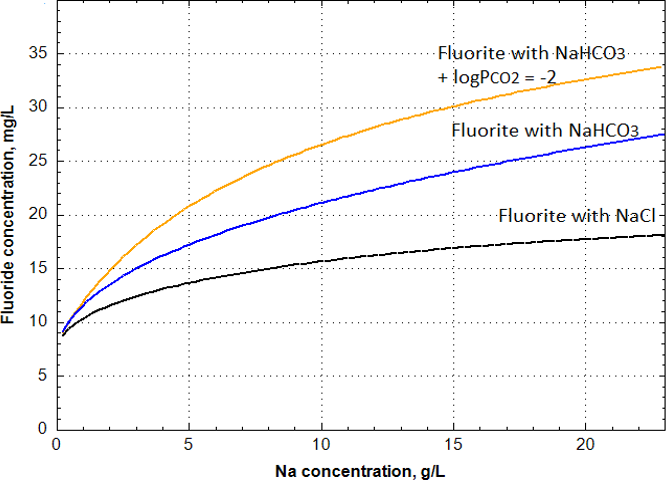
Figure 5 – Fluorite solubility (in terms of F– concentration) with increasing concentrations of NaHCO3 (blue line) and with a constant partial pressure of CO2(g) (yellow line) compared to NaCl (black line).
If we consider fluorite and fluorapatite solubility in solutions with dissolved inorganic carbon (DIC), the solubility increases much the same as in NaCl solutions. However, with increasing HCO3 concentration, the solution becomes saturated with respect to calcite and the F concentration increases substantially, depending on the partial pressure of CO2 (PCO2). Precipitation of calcite decreases the Ca ion concentration and activity, thereby increasing F concentration because of fluorite solubility equilibrium and the law of mass action as illustrated in Equation 9.
| CaF2 ↔ Ca2+ + 2F− log Ksp = −10.6 | (9) |
If calcite and fluorite are both at equilibrium solubility, then the reaction is as shown in Equation 10.
| CaF2 + HCO3− → CaCO3 + 2F− + H+ | (10) |
However, if HCO3– is being contributed from an additional source such as organic carbon oxidation, then more calcite will precipitate (depending on pH), and the F concentration will continue to increase accordingly. Calcite saturation and precipitation is then a requirement for elevating F concentrations and sources of additional bicarbonate alkalinity can promote calcite precipitation. Fluorapatite solubility is similarly affected by bicarbonate ion and accompanied by calcite precipitation which decreases with increasing PCO2, but the F concentrations are substantially lower, and dissolution is often incongruent. Groundwater in many aquifers increases in alkalinity with increasing age and interaction with: organic matter decomposition in the soil zone, organic matter decomposition in buried sediments, and CO2 gas rising from thermal sources.
